Do you think you know everything there is to know about nuclear reactors? Well, buckle up because this article is about to blow your mind. We’re diving into the world of U-235 fission products – those radioactive byproducts that come from the splitting of uranium-235. Trust me, these little guys are not something you want to mess with. They can be highly hazardous and have a big impact on our environment. So, let’s strap in and learn all about these fission products and the challenges they pose for disposal.
Formation and Release of Fission Products
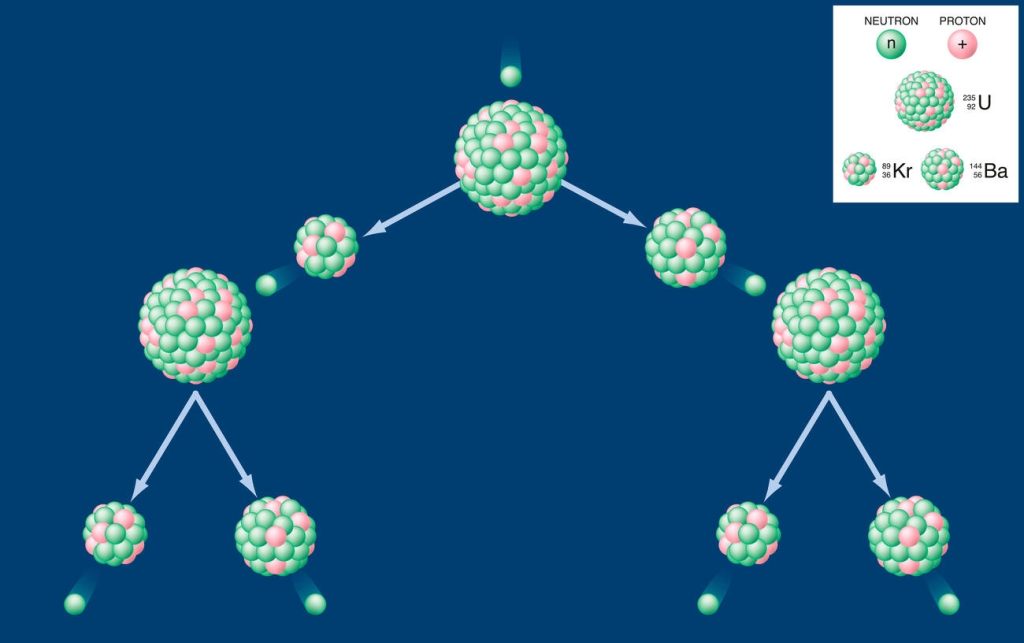
You should be aware that fission products are formed and released during the process of uranium-235 fission. When uranium-235 undergoes fission, it splits into two smaller atomic nuclei, releasing a large amount of energy. These smaller atomic nuclei are known as fission products. They can include various isotopes of elements such as cesium, iodine, strontium, and xenon. The formation and release of these fission products occur due to the impact of high-energy neutrons on the uranium-235 nucleus. As the uranium-235 nucleus absorbs a neutron, it becomes unstable and splits apart, producing new atoms in the process. These newly formed atoms then rapidly decay into different isotopes through radioactive decay, releasing additional radiation in the form of gamma rays and beta particles. It is important to understand this process in order to properly manage and handle radioactive waste produced by nuclear reactors.
Environmental Impact and Mobility of Fission Products
The release of fission byproducts during nuclear accidents and weapons production can have significant environmental impacts. Nuclear fission, the process of splitting uranium atoms, is responsible for these byproducts. When uranium undergoes fission in a nuclear reactor or during a nuclear reaction, it releases energy along with smaller atomic number atoms and highly radioactive substances. Some important fission products include iodine-129, cesium-137, strontium (Sr), technetium (Tc), selenium (Se), and zirconium (Zr). These radioactive elements can contaminate the environment and pose long-term disposal problems due to their long half-lives. The mobility of fission products depends on their chemical properties and physical form of contamination. Therefore, it is crucial to properly manage and dispose of these hazardous materials to minimize their impact on the environment.
Analysis and Measurement of Fission Products
When analyzing and measuring fission products, it’s important to employ techniques such as delayed neutron counting, sequential radiochemical methods, and laser fluorometric methods. These methods allow scientists to accurately determine the presence and concentration of specific fission products in various materials. In the field of nuclear physics, these techniques are crucial for understanding the behavior of uranium-235 and other fissile materials in reactors. By studying the fission process and its byproducts, scientists can gain insights into how much energy is produced through nuclear fission and develop safer and more efficient reactor designs. The starting materials for nuclear fission are typically uranium-235 or plutonium-239, which undergo a chain reaction when bombarded with neutrons. Through careful analysis using these techniques, researchers can better understand the complexities of fission products and their impact on nuclear reactions.
Fission Products in Nuclear Power Plants
Gaseous fission products, such as iodine, krypton, and xenon, are commonly produced during operation of nuclear power plants. These fission products pose challenges for long-term disposal due to their high radioactivity and hazardous nature. To manage them effectively, nuclear power plants employ certain techniques:
1) Activated carbon beds: These beds are used to trap the gaseous fission products and reduce their concentration in the coolant gas. Nuclear grade activated carbons made from coconut shell or coal-based precursors are used for this purpose.
2) Liquid nitrogen-cooled activated charcoal beds: These beds are utilized to trap stable gases and impurities. The cooling with liquid nitrogen helps maintain a low temperature which aids in capturing these gases effectively.
3) Decay time: The adsorption beds have long contact times to allow radioactive gases to decay before they are released into the environment.
These methods help ensure that the release of gaseous fission products is minimized and controlled during the operation of nuclear power plants.
Challenges in Nuclear Waste Disposal of Fission Products
Managing the disposal of fission products from nuclear waste poses significant challenges in ensuring long-term safety and containment. You must carefully consider the radioactive nature of these byproducts and their potential impact on the environment. The fission products, such as iodine-129 and cesium-137, are highly radioactive and hazardous. They have long half-lives, which means they remain dangerous for many years to come. Proper containment and isolation are crucial to prevent these substances from leaking into the surrounding environment. This requires designing and constructing robust storage facilities that can withstand corrosion, mechanical failure, and water contact. Additionally, finding suitable geological formations for deep repository sites is essential to minimize any potential release of radionuclides into groundwater or other natural systems. Meeting these challenges is vital to ensure the safe management of fission products and protect both human health and the environment.
Fission Product Transformation and Transmutation
Now let’s explore the topic of Fission Product Transformation and Transmutation. In this subtopic, you will learn about how fission products can be transformed into different forms through various processes. Here are three key points to consider:
- Charged particle irradiation: This method involves exposing fission products to high-energy charged particles, which can transform them into shorter-lived or stable elements. However, this process is expensive.
- Neutron irradiation: Alternatively, neutron irradiation can also be used to transform fission products. Intense accelerator-driven spallation neutron sources have been proposed for this purpose. While neutron irradiation produces almost as many new fission products as it destroys, it still offers potential for transmutation.
- Controlled thermonuclear reactors (CTR): CTRs have the capability to transform certain fission products like strontium (Sr). However, they are less efficient when it comes to transforming other fission products like cesium-137 (Cs-137).